About Us
Our History

A vision, realized
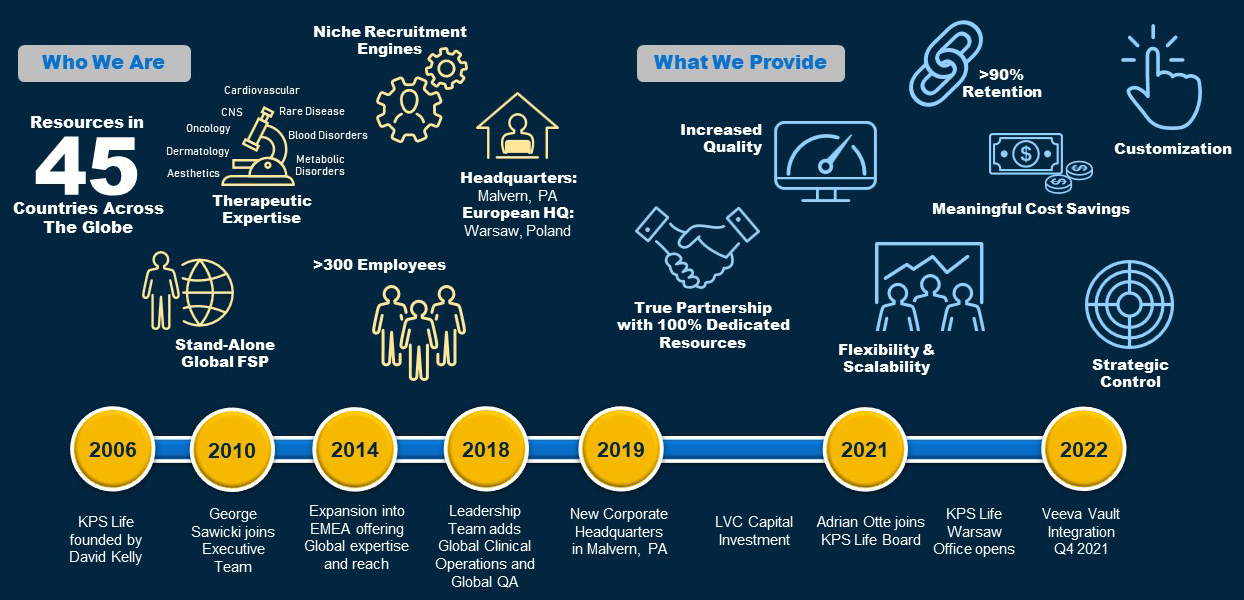
In 2006, after almost twenty years in the pharmaceutical industry, David Kelly, having grown frustrated with the full-service outsourcing (FSO) model, left the sector with a vision to start his own company: one whose mission was to ensure the quality and success of clinical trials — with the best clinical research professionals, and peerless oversight — and do so at a fraction of what large FSOs charge. In short, he sought to create a functional service provider model that would redefine the very concept: one dedicated to quality outcomes, best-in-class embedded resources, customer centricity, leading technologies, and unmatched speed and responsiveness.
Today, KPS Life is a global industry leader, with operations on six continents and practically every country, working with some of the most well-known and respected brands in the industry.